Spine Fractures Explained: What Medical Experts Want You to Know
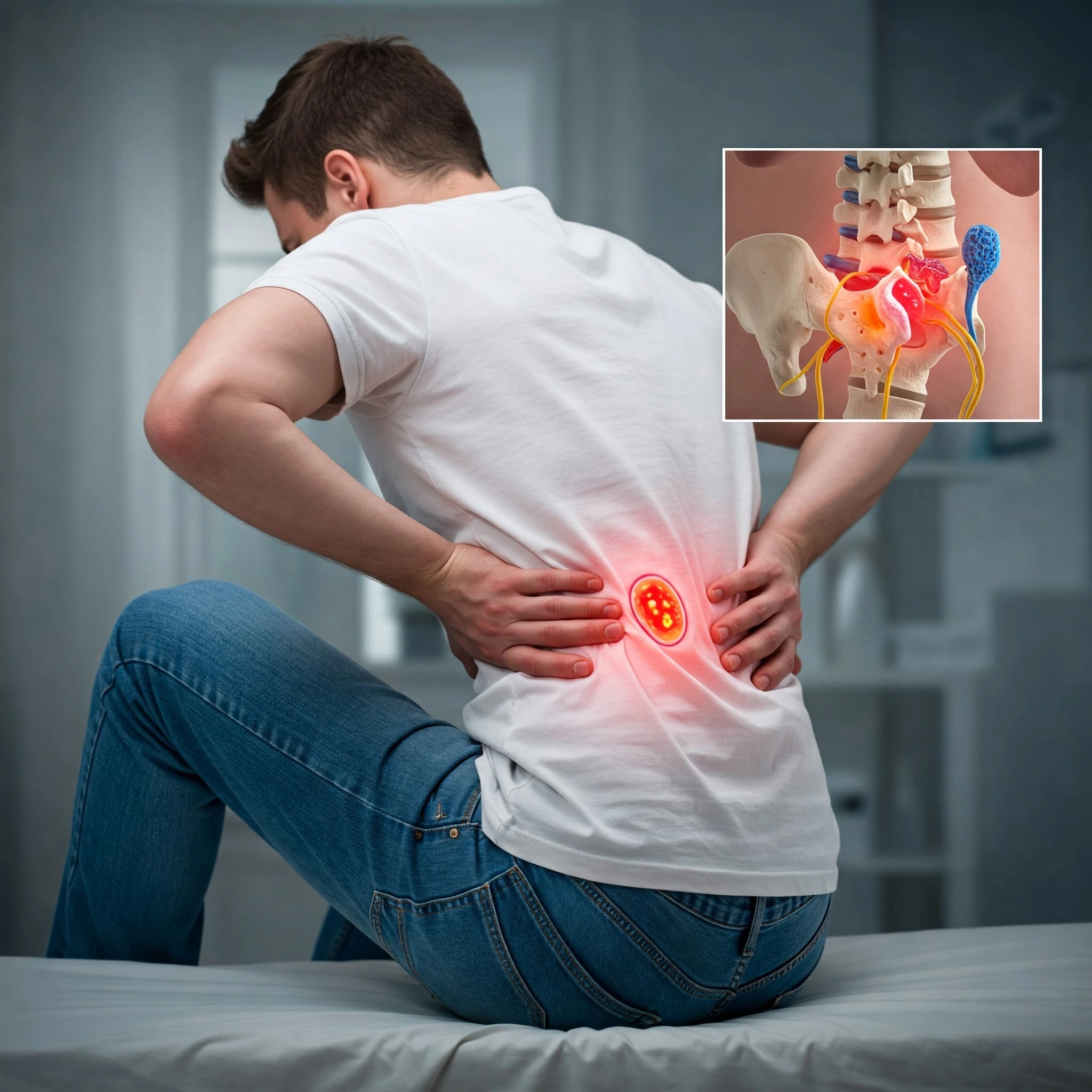
Spine fractures affect approximately 1.5 million Americans each year, with many cases going undiagnosed until serious complications develop. These potentially debilitating injuries can dramatically alter a person’s quality of life, causing chronic pain, limited mobility, and in severe cases, permanent neurological damage.
Although vertebral fractures can occur throughout the spinal column, they most commonly manifest as vertebral compression fractures in the thoracic and lumbar regions. Specifically, thoracic compression fractures and lumbar compression fractures account for nearly 75% of all cases, often resulting from osteoporosis, trauma, or underlying pathological conditions. Treatment options vary widely depending on fracture severity and patient health, ranging from conservative management to interventional procedures like kyphoplasty.
This comprehensive guide examines the anatomical basis of spine fractures, classification systems, diagnostic approaches, and treatment options. Additionally, we’ll explore potential complications and prognosis factors to help you better understand these common yet complex injuries. Whether you’re a patient recently diagnosed with a vertebral fracture or a family member seeking information, this article provides essential knowledge from medical experts in spine health.
Anatomical Basis of Spine Fractures
The vertebral column serves as the central support structure for the human body, housing and protecting the spinal cord while enabling movement. Understanding its anatomical components provides crucial insight into why and how spine fractures occur.
Vertebral Column Structure: Body, Arch, and Ligaments
The vertebral column consists of 33 stacked vertebrae forming the spine from the base of the skull to the tailbone [1]. Each vertebra shares a common structural foundation comprising an anterior vertebral body and a posterior vertebral arch that together form the vertebral foramen—a protective canal for the spinal cord [2]. The vertebral bodies primarily bear weight, consequently growing larger in the lower regions to support increased loads [2].
Between adjacent vertebrae lie intervertebral disks that function as shock absorbers and permit flexibility. These disks consist of a soft, gel-like nucleus pulposus surrounded by a tough, fibrous outer ring called the annulus fibrosus [1]. Several key ligaments reinforce the vertebral column:
-
- The anterior longitudinal ligament runs along the front of the vertebral bodies from skull to sacrum, limiting extension
-
- The posterior longitudinal ligament travels inside the vertebral canal along the posterior surface of the vertebral bodies, preventing hyperflexion and disk herniation
-
- Ligamenta flava connect adjacent laminae, preventing sudden separation during flexion
-
- Interspinous and supraspinous ligaments join the spinous processes of adjacent vertebrae [2]
These ligamentous structures provide stability while still allowing controlled movement throughout the spine.
Load Distribution and Axial Compression Mechanics
During daily activities, the vertebral column experiences various forces, with axial compression being particularly significant in fracture development. Research shows that standing and walking generate approximately 1000 N of compression through spinal musculature, while lifting activities can produce several thousand Newtons of force [3]. The normal vertebral structure efficiently distributes these loads, but excessive forces can overwhelm this system.
Notably, the lumbar spine supports the upper body’s weight and bears most of the stress from lifting and carrying objects [1]. During activities that shift body weight forward—such as trunk flexion or carrying weights in front—compressive loads increase dramatically at certain vertebral levels [4]. Moreover, studies reveal that increased thoracic kyphosis (forward curve) accentuates peak loading in the thoracolumbar region [4].
When axial compression exceeds the bone’s structural integrity, vertebral fractures occur. This typically results from excessive downward pressure on the vertebral body, often combined with forward bending [5]. Consequently, the cancellous bone inside the vertebral body crushes or compresses, forming a characteristic wedge shape.
Common Fracture Sites: T11-L2 and C1-C2
Research consistently demonstrates that 60-70% of thoracolumbar fractures occur in the T11-L2 region [6]. This biomechanically vulnerable area exists at the junction between the relatively rigid thoracic spine and the more mobile lumbar region. The thoracolumbar junction experiences higher mechanical loading during daily activities compared to other spinal areas [4].
The T11-L1 region shows consistently higher stress concentrations, with studies identifying peak factor-of-risk values (ratio of compressive load to vertebral strength) at T11 for 83% of tested activities [4]. Furthermore, increased thoracic kyphosis heightens this concentration of stress [4]. The underlying mechanism appears related to selective activation of multifidus muscle fascicles in the thoracolumbar region, significantly contributing to compressive loading [4].
In the cervical region, the C1-C2 vertebrae (atlas and axis) represent another common fracture site due to their unique structure. Unlike other vertebrae, they lack vertebral bodies and instead feature specialized components that permit greater rotation but offer less stability under certain forces [2].
Classification and Causes of Vertebral Fractures
Vertebral fractures encompass a range of injuries with distinct patterns, causes, and treatment considerations. Proper classification of these fractures provides a framework for clinical decision-making and helps predict outcomes.
Osteoporotic Compression Fractures
Osteoporotic compression fractures represent the most common type of vertebral fracture, with an estimated 700,000 cases occurring annually in the United States [7]. These fractures primarily affect the vertebral body—the thick, rounded anterior portion of each vertebra. Initially, many patients remain unaware of these injuries, as only about one-third receive clinical diagnosis [7].
Characterized by reduced bone mass and architectural deterioration, osteoporosis weakens the vertebrae, making them susceptible to fracture even during routine activities such as coughing, sneezing, or getting out of a car [8]. The vertebral body collapses under pressure, typically creating a wedge-shaped deformity [8].
The consequences of these fractures extend beyond immediate pain, often including height loss, kyphosis (forward curvature of the spine), restrictive lung disease, and reduced quality of life [7]. Importantly, a single spine fracture increases the risk of subsequent fractures dramatically—fivefold for another spine fracture and twofold for hip and other fractures [7].
Traumatic Burst and Flexion-Distraction Injuries
In contrast to osteoporotic fractures, traumatic spine fractures number approximately 160,000 annually, with 50% affecting the thoracolumbar junction [9]. These injuries typically result from high-energy events such as falls, sports accidents, or motor vehicle collisions.
Burst fractures occur when extreme axial force crushes the vertebra, causing it to break in multiple places [8]. Unlike simple compression fractures, burst fractures involve both anterior and posterior elements of the vertebra with bone fragments potentially entering the spinal canal [10]. These injuries frequently result from falling from height and landing in an upright position [11].
Flexion-distraction injuries (also called Chance fractures) develop through a different mechanism—when the spine is forcibly bent forward over a fulcrum, such as a seatbelt during sudden deceleration [1]. These horizontal fractures extend from posterior to anterior through the spinous process, pedicles, and vertebral body [1]. Clinically significant, flexion-distraction injuries carry a high association with abdominal injuries—up to 50% of cases involve concurrent intra-abdominal trauma [1].
Pathological Fractures from Metastasis or Infection
Pathological fractures develop through areas of bone already weakened by disease processes. Metastatic cancer represents a significant cause, with bone being the most common site for metastasis in breast cancer patients—affecting up to 70% of women with metastatic breast cancer [12].
These fractures substantially impact quality of life, with approximately 68% of patients with skeletal metastasis experiencing pain, and 10-20% of those with long-bone metastases eventually sustaining pathological fractures [12]. Especially noteworthy, in 3-4% of cases, pathological fracture serves as the first indication of underlying cancer [12].
Other conditions that predispose to pathological fractures include infections, multiple myeloma, hyperthyroidism, and long-term steroid use [9]. Early intervention for patients with metastatic bone disease reduces both morbidity and overall healthcare costs [12].
TLICS and SLICS Scoring Systems Overview
The Thoracolumbar Injury Classification and Severity score (TLICS) and Subaxial Cervical Injury Classification and Severity scale (SLICS) help standardize assessment and guide treatment decisions [9].
TLICS evaluates three key components: injury morphology, posterior ligamentous complex integrity, and neurological status [13]. Each component receives a point value based on severity, with the total score guiding treatment decisions—scores of 5 or higher generally indicate surgical intervention [9].
Similarly, SLICS assesses subaxial cervical spine injuries (C3-C7) through three categories: morphologic features, integrity of the discoligamentous complex, and neurological status [14]. The scoring system ranges from 0 (least severe) to 4 (most severe) for each category, with treatment recommendations based on the cumulative score [14].
Both classification systems aim to facilitate communication between physicians and predict which patients will benefit most from surgical versus non-surgical management [14]. Nevertheless, studies have shown variable inter-observer reliability, suggesting limitations in their clinical application [14].
Clinical Presentation and Diagnostic Workflow
Diagnosing spine fractures demands careful attention to pain characteristics and neurological symptoms, followed by appropriate imaging selection. The clinical presentation varies substantially depending on fracture etiology, location, and patient factors.
Acute vs Chronic Pain Patterns in VCFs
Pain manifestations in vertebral compression fractures (VCFs) follow distinct patterns that aid diagnosis. Remarkably, despite the common prevalence of these fractures, only about one-third cause severe pain warranting medical attention [4]. Acute fractures typically present with sudden, severe, and focal pain at the level of injury that worsens with movement and improves with rest [15].
Pain distribution often follows identifiable patterns, as research has categorized them into three main types [4]:
-
- Type A: Localized paravertebral pain centered at the midline
-
- Type B: Diffuse paravertebral pain, either confined to the posterior back (B1) or extending to the anterior chest (B2)
-
- Type C: Remote low back or lumbosacral pain distant from the actual fracture site
Beyond location, timing offers diagnostic clues. Patients with chronic VCFs frequently experience muscle fatigue pain from compensatory posture changes after height loss [16]. Furthermore, compression fractures can sometimes remain asymptomatic until detected incidentally during imaging for other conditions [15].
Neurological Deficit Assessment using ASIA Scale
Neurological evaluation following spine fractures relies primarily on the American Spinal Injury Association (ASIA) Impairment Scale. This standardized assessment categorizes spinal cord injury severity into five grades [17]:
-
- Grade A: Complete injury with no sensory or motor function preserved in sacral segments S4-5
-
- Grade B: Incomplete injury with preserved sensory but not motor function below the injury level
-
- Grade C: Incomplete injury with preserved motor function where most key muscles below the injury have strength less than grade 3
-
- Grade D: Incomplete injury with preserved motor function where most key muscles below the injury have strength grade 3 or higher
-
- Grade E: Normal sensory and motor functions
Accordingly, this classification helps predict recovery potential, with approximately 30-40% of individuals initially classified as ASIA B converting to ASIA C or D within the first year [18].
Imaging Modalities: CT, MRI, and DEXA Scan
Diagnostic imaging begins with plain radiography, typically anteroposterior and lateral views, which can identify most vertebral fractures [19]. Subsequently, computed tomography (CT) provides superior visualization of fracture morphology in three planes and has become indispensable for thorough fracture evaluation [19].
Magnetic resonance imaging (MRI) excels at revealing soft tissue involvement and distinguishing between acute and chronic fractures through signal intensity characteristics [6]. Primarily, MRI helps identify ligamentous injuries, neurological compression, and potential malignancy [19].
For osteoporosis assessment, dual-energy X-ray absorptiometry (DEXA) serves as the gold standard [9]. Nonetheless, DEXA has limitations in patients with degenerative changes or aortic calcifications, who often comprise the spine surgery population [3].
Genant Semiquantitative Grading for Osteoporotic VCFs
The Genant semiquantitative method provides standardized classification for vertebral compression fractures based on height reduction [20]. This widely adopted system categorizes fractures into:
-
- Grade 0: Normal vertebral body
-
- Grade 1: Mild deformity (20-25% height reduction)
-
- Grade 2: Moderate deformity (26-40% height reduction)
-
- Grade 3: Severe deformity (>40% height reduction)
The assessment evaluates vertebral bodies from T4 to L4, considering both height reduction and morphological changes [20]. This system demonstrates high reliability, with intraobserver agreement of 93-99% and interobserver agreement of 90-99% [20].
First thing to remember, any reduction in vertebral height of 20% or more, or at least 4mm compared to baseline height, indicates a positive diagnosis for compression fracture [16].
Treatment Modalities and Surgical Indications
Effective management of spine fractures requires a tailored approach based on fracture stability, neurological status, and patient factors. Treatment strategies range from conservative care to complex surgical interventions, with the primary goal of relieving pain while preserving or restoring spinal stability.
Conservative Management: Bracing and Analgesics
Most vertebral compression fractures (VCFs) respond well to non-surgical treatments. Bracing typically involves thoracolumbar sacral orthosis (TLSO) or Jewett braces, worn for 4-12 weeks until pain resolves and radiographic healing occurs [6]. Indeed, studies show that bracing provides significant benefits for trunk muscle strength, posture, and quality of life [5]. However, potential drawbacks include inadequate immobilization, skin sores, decreased pulmonary function, and core muscle weakness [5].
For pain control, NSAIDs and acetaminophen often provide sufficient relief [21]. Research indicates that calcitonin (standardized mean difference −4.86; 95% CI, −6.87 to −2.86) and NSAIDs (SMD, −3.94; 95% CI, −7.30 to −0.58) demonstrate benefits for short-term pain management compared to placebo [22]. For osteoporotic fractures, teriparatide has shown excellent results in increasing vertebral bone mineral density [22].
Vertebroplasty vs Kyphoplasty: Indications and Outcomes
Vertebroplasty and kyphoplasty are minimally invasive procedures for patients with severe, persistent pain. Vertebroplasty stabilizes fractures through percutaneous cement injection without significantly restoring vertebral height [6]. In contrast, kyphoplasty uses an inflatable balloon to partially restore vertebral height, offering superior correction of segmental kyphosis and reduced cement leakage rates [6].
Clinical data show that patients undergoing vertebral augmentation procedures experience pain relief almost 3 months faster than those receiving conservative treatment [23]. Furthermore, the progression or re-fracture rate is significantly lower with surgical intervention (4.8% vs 29.7% in conservative treatment) [23].
Surgical Stabilization for Unstable Fractures
Surgical stabilization becomes necessary for unstable fractures, neurological compromise, or significant kyphotic deformity. The AO Spine Thoracolumbar Injury Classification system guides treatment decisions based on fracture morphology, neurological status, and clinical modifiers [6].
For severe osteoporotic fractures (OF 4-5), short-segment posterior instrumentation often supplemented with vertebral augmentation is recommended [6]. Early surgical intervention (within 72 hours) for unstable thoracolumbar fractures significantly decreases hospital length of stay compared to delayed fixation [24].
Management of Spinal Cord Injury with MAP > 85
For acute traumatic spinal cord injuries, maintaining mean arterial pressure (MAP) above 85 mmHg for 7 days is the standard practice to enhance spinal cord perfusion [2]. This recommendation, albeit based on Class III evidence, aims to improve neurological outcomes [2].
Studies demonstrate that higher proportions of MAP measurements ≥85 mmHg correlate with better neurological improvement [25]. Practically, approximately 78% of patients require vasopressors to achieve this target [26]. Recent research suggests spinal cord perfusion pressure (SCPP) >50 mmHg may be an even stronger predictor of neurological recovery than MAP alone [27].
Complications and Prognostic Indicators
Complications following spine fractures can profoundly affect quality of life and survival. Understanding these potential outcomes helps guide treatment decisions and set realistic expectations.
Kyphotic Deformity and Segmental Instability
Vertebral fractures fundamentally alter spine biomechanics, primarily through loss of height in the vertebral body and disruption of the posterior tension band [28]. As kyphosis progresses, compensatory mechanisms attempt to maintain sagittal balance, yet many patients develop sagittal imbalance as loads on the anterior column increase [29]. In essence, this progressive kyphosis stretches paraspinal muscles, causing inflammation, pain, and eventually neurological damage through cauda equina fiber stretching [28]. Short-segment fixation carries a high pseudoarthrosis rate (9-54%) [29], certainly contributing to progressive kyphotic deformity. Furthermore, studies reveal that implant removal significantly increases kyphosis development risk [29].
Neurological Compromise and Spinal Stenosis
Delayed neurological deficits represent serious complications, occurring in 12.1% of vertebral fractures [11]. For instance, bilateral foraminal stenosis can develop when the caudal part of a vertebral body collapses, causing radiculopathy even without direct cord compression [11]. Research demonstrates a positive correlation between the extent of cervical and thoracic spinal canal compression and neurological deficit severity [30]. Notably, even cases without initial neurological symptoms may develop progressive deficits from bone fragment retropulsion, pseudoarthrosis formation, or local kyphosis [11].
Mortality Risk in Untreated Osteoporotic VCFs
Osteoporotic vertebral compression fractures significantly increase mortality compared to age-matched controls [6]. Specifically, survival rates decrease to 53.9% at 3 years, 30.9% at 5 years, and merely 10.5% at 7 years [6]. Correspondingly, a population-based cohort study found a 22% higher mortality risk in OVCF patients versus controls [31]. Risk factors include male sex, advanced age, low income, and comorbidities—with congestive heart failure carrying the highest risk (HR 2.26) [32]. The mortality mechanism appears related to prolonged immobility leading to pulmonary disease, deep vein thrombosis, and exacerbated comorbidities [32].
Cement Leakage and Hardware Failure Risks
Vertebral augmentation procedures, albeit beneficial, carry specific risks. Cement leakage occurs frequently yet remains typically asymptomatic [6]. Nonetheless, severe complications may arise from cement extravasation into the epidural space, neural foramina, or venous system [7]. Pulmonary cement embolism, though rare, can be fatal [8]. Adjacent vertebral fractures develop in approximately 10-40% of cases following augmentation [8], frequently within months of the procedure. Risk factors for revision surgery include female sex, diabetes mellitus, split-type fracture, large angular motion, and large endplate deficit [8].
Conclusion
Spine fractures remain a significant health concern affecting approximately 1.5 million Americans annually. Therefore, understanding their anatomical basis, classification, diagnosis, and treatment options becomes crucial for both patients and healthcare providers. The thoracolumbar junction (T11-L2) proves particularly vulnerable due to the biomechanical stress concentration at this transition between the rigid thoracic and mobile lumbar regions.
Osteoporotic compression fractures undoubtedly represent the most common type, often developing from weakened bone structure that collapses during routine activities. Meanwhile, traumatic fractures typically result from high-energy events, creating burst or flexion-distraction injuries with potentially devastating consequences. Additionally, pathological fractures stemming from metastatic cancer or infection require special consideration during diagnosis and treatment planning.
The diagnostic workflow for spine fractures relies on careful assessment of pain patterns and neurological deficits, followed by appropriate imaging. Consequently, CT scans provide detailed fracture morphology, whereas MRI excels at revealing soft tissue involvement and distinguishing between acute and chronic fractures. The Genant semiquantitative method subsequently offers standardized classification based on vertebral height reduction.
Treatment approaches range from conservative management with bracing and analgesics to minimally invasive procedures like vertebroplasty and kyphoplasty. For unstable fractures or those with neurological compromise, surgical stabilization becomes necessary. Nevertheless, complications may still arise, including kyphotic deformity, neurological compromise, and hardware failure.
The impact of vertebral fractures extends beyond immediate pain. Patients with untreated osteoporotic vertebral compression fractures face significantly increased mortality rates compared to age-matched controls. Thus, early diagnosis and appropriate intervention remain essential for optimizing outcomes and maintaining quality of life. Through increased awareness and prompt treatment, patients can potentially avoid the progressive deformity, chronic pain, and functional limitations that often accompany these common yet complex spinal injuries.
References
[1] – https://www.ncbi.nlm.nih.gov/books/NBK536926/
[2] – https://pmc.ncbi.nlm.nih.gov/articles/PMC8081322/
[3] – https://pmc.ncbi.nlm.nih.gov/articles/PMC10119682/
[4] – https://pmc.ncbi.nlm.nih.gov/articles/PMC2610635/
[5] – https://pmc.ncbi.nlm.nih.gov/articles/PMC9827207/
[6] – https://www.ncbi.nlm.nih.gov/books/NBK448171/
[7] – https://www.mdpi.com/1648-9144/59/9/1536
[8] – https://www.e-neurospine.org/journal/view.php?number=1454
[9] – https://www.ncbi.nlm.nih.gov/books/NBK547673/
[10] – https://orthoinfo.aaos.org/en/diseases–conditions/fractures-of-the-thoracic-and-lumbar-spine/
[11] – https://pmc.ncbi.nlm.nih.gov/articles/PMC3603666/
[12] – https://www.ccjm.org/content/89/7/393
[13] – https://radiologyassistant.nl/neuroradiology/spine/tlics-classification
[14] – https://pmc.ncbi.nlm.nih.gov/articles/PMC7491911/
[15] – https://nyulangone.org/conditions/spine-compression-fractures/diagnosis
[16] – https://www.aafp.org/pubs/afp/issues/2004/0101/p111.html
[17] – https://emedicine.medscape.com/article/2172437-overview
[18] – https://nascic.org/the-asia-impairment-scale/
[19] – https://emedicine.medscape.com/article/398102-overview
[20] – https://pmc.ncbi.nlm.nih.gov/articles/PMC3591834/
[21] – https://my.clevelandclinic.org/health/diseases/17498-spinal-fractures
[22] – https://pmc.ncbi.nlm.nih.gov/articles/PMC11380106/
[23] – https://bmcmusculoskeletdisord.biomedcentral.com/articles/10.1186/s12891-023-07041-1
[24] – https://www.sciencedirect.com/science/article/pii/S2772632025000091
[25] – https://pubmed.ncbi.nlm.nih.gov/33003016/
[26] – https://www.sciencedirect.com/science/article/pii/S1036731424002820
[27] – http://e-neurospine.org/upload/pdf/ns-2040144-072.pdf
[28] – https://pmc.ncbi.nlm.nih.gov/articles/PMC8752698/
[29] – https://www.e-neurospine.org/upload/pdf/ns-2142340-170.pdf
[32] –https://pmc.ncbi.nlm.nih.gov/articles/PMC10501656/