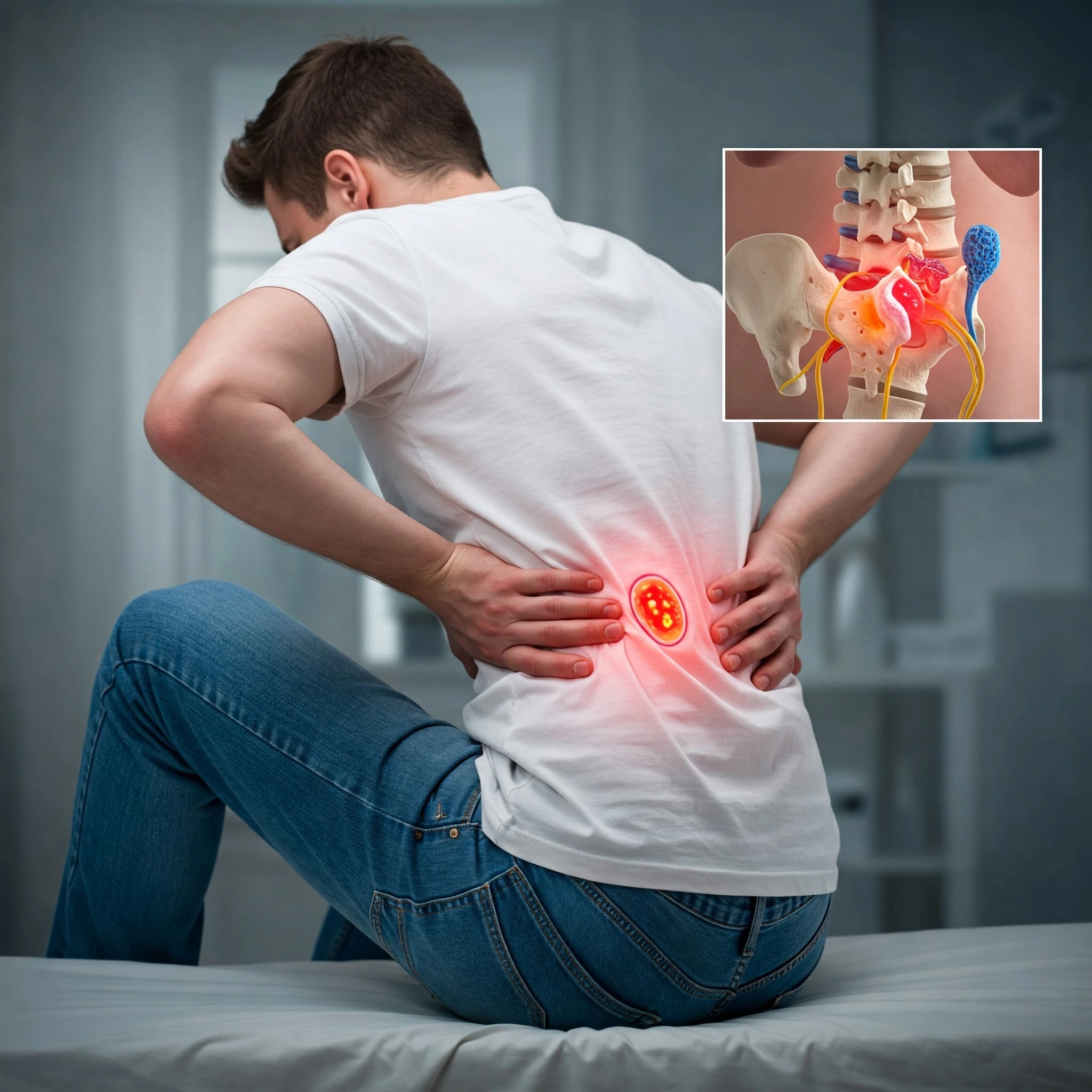
Osteoarthritis (OA) is no longer seen only as “wear and tear.” In its early stages there’s a window of opportunity: inflammation is lower, cartilage loss is incomplete, and the joint environment is still responsive to therapies that may slow — or even partially reverse — damage. While PRP and traditional stem-cell (MSC) injections remain widely discussed, a new generation of regenerative treatments is emerging: exosomes and cell-free biologics, micro-fragmented adipose tissue (MFAT), bone marrow aspirate concentrate (BMAC) refinements, peptide-based scaffolds and hydrogels, and even gene-therapy approaches. This post explains these options, summarizes current evidence, and gives practical guidance for clinicians and patients considering early OA interventions.
Why focus on early OA?
Early OA (often Kellgren-Lawrence grade I–II) features cartilage softening, mild fibrillation, synovial inflammation, and pain that is often activity-related. Intervening early aims to:
- Reduce inflammatory signaling that perpetuates cartilage loss.
- Preserve remaining cartilage and subchondral bone architecture.
- Delay or avoid joint replacement.
Multiple reviews emphasize that biologic therapies have the greatest chance to alter disease trajectory if applied before extensive structural breakdown.
How these new approaches differ from PRP / simple MSC injections
PRP is a concentrated source of growth factors; MSC injections introduce cells thought to modulate inflammation and help tissue repair. Newer strategies either improve the cell product, deliver cell-derived factors without live cells, or combine biologics with engineered scaffolds to improve retention and tissue regeneration. Important advantages include lower immune risk (cell-free options like exosomes), higher local payload/retention (scaffolds, hydrogels), and delivery of specific therapeutic genes or peptides for sustained effect.
Emerging options (what they are + current evidence)
1. Exosome therapy (cell-free, paracrine-factor delivery)
What: Exosomes are nano-sized vesicles released by cells (often MSCs or adipose-derived stem cells). They carry proteins, miRNAs, and signaling molecules that modulate inflammation and stimulate matrix synthesis.
Why promising: They deliver the beneficial paracrine signals of MSCs without transplanting living cells — fewer safety worries (e.g., tumorigenicity) and easier standardization. Preclinical data show cartilage protection and reduced synovitis; early human/clinical feasibility studies and systematic reviews conclude exosomes are a promising next-gen therapy but require standardized manufacturing and dosing studies.
Clinical takeaway: Excellent translational promise; several groups running early-phase trials. Not yet a routine, off-the-shelf option in most practices.
2. Micro-fragmented adipose tissue (MFAT / SVF)
What: MFAT is mechanically processed fat containing a stromal vascular fraction (SVF) — a mix of MSCs, pericytes, growth factors and extracellular matrix. It is prepared at point-of-care, requiring only lipoaspirate and minimal processing.
Evidence: Multiple clinical series and some comparative studies report pain and function improvements up to 12 months, with MFAT often showing durable benefit in moderate OA; randomized data are still limited but growing. MFAT may provide a scaffold + cellular milieu that sustains repair signals longer than simple PRP.
Clinical takeaway: Practical for clinics already offering adipose-derived treatments. Good option for early–moderate OA where joint space and alignment are reasonable.
3. Bone marrow aspirate concentrate (BMAC) — refinements and evidence
What: BMAC concentrates bone marrow cells, platelets, and growth factors; it’s often used for intra-articular injections or surgical augmentation.
Evidence: Systematic reviews and RCTs show BMAC can provide pain relief and improved function in knee OA, sometimes outperforming hyaluronic acid and showing comparable short-term benefit to PRP in certain trials. Heterogeneous preparation and dose remain challenges; recent reviews urge standardized protocols to clarify which patients benefit most.
Clinical takeaway: Reasonable evidence base for symptomatic improvement; best used with standardized harvest/processing protocols and realistic expectations about structural regeneration.
4. Peptide-enhanced scaffolds & injectable hydrogels (the “injectable goo”)
What: Engineered hydrogels and peptide-based scaffolds can act as both a physical matrix and a biologic delivery vehicle (e.g., for growth factors, peptides, or cells). Some recent preclinical work shows these materials can support cartilage regrowth when paired with chondroinductive cues. Notable examples include hyaluronic-acid/peptide hydrogels that promote matrix deposition.
Clinical takeaway: Most data are preclinical or early translational. These technologies show potential to actually regrow cartilage when combined with correct biologic signals, but robust human trial data are still forthcoming.
5. Gene therapy for joints (sustained intra-articular biologic expression)
What: Intra-articular gene therapy uses viral or non-viral vectors to deliver genes that code for anti-inflammatory proteins (e.g., IL-1Ra), growth factors, or enzymes that modulate matrix turnover — providing sustained local production from within the joint.
Progress: Clinical pipelines and some early human trials have demonstrated feasibility of achieving sustained transgene expression in joints; vector safety and control of expression are central research focuses. Reviews emphasize promising early results but caution more long-term safety and efficacy data are needed.
Clinical takeaway: High potential for disease-modifying therapy, but still largely experimental and offered through clinical trials.
Combining strategies: the likely future
Hybrid approaches are already under study: MFAT or BMAC combined with peptide hydrogels to improve retention; exosome-loaded scaffolds; or gene-activated scaffolds that release therapeutic proteins locally. Combining a cell or cell-product with a scaffold and targeted biologic (peptide or gene) addresses three problems at once: payload, retention, and sustained signaling. Reviews in tissue engineering emphasize such multifunctional constructs as a leading direction.
Who is a good candidate for these emerging treatments?
- Early OA (KL grade I–II, sometimes early III) with focal pain and preserved joint space.
- Patients who have failed conservative care (exercise, weight loss, analgesics) but want to avoid or defer arthroplasty.
- No active infection, well-controlled comorbidities, and realistic expectations — these therapies often improve pain and function and in some cases slow progression, but they are not guaranteed “cures.”
Safety, regulation, and practical considerations
- Standardization matters. Outcome variability often stems from differences in harvesting, processing, and dosing. Choose suppliers/labs with transparent protocols and quality metrics.
- Regulatory status varies. Many of these therapies are experimental or available under limited regulatory pathways; gene therapies and some exosome products typically require clinical-trial enrollment.
- Adverse events reported are usually minor (injection pain, transient swelling); long-term safety (especially with viral vectors and novel biomaterials) needs ongoing surveillance.
Practical clinic workflow (example)
- Confirm early stage via imaging and exam; rule out mechanical/instability causes.
- Optimize non-operative measures (exercise, weight loss, gait/orthotics).
- Discuss options: PRP, BMAC, MFAT, exosomes (trial setting), or combined scaffold approaches. Explain evidence level and costs.
- If proceeding, use standardized consent + document baseline PROMs (e.g., KOOS, WOMAC). Schedule objective follow-up at 3, 6, 12 months.
FAQs
Q: Are exosomes the same as stem cells?
No — exosomes are cell-derived vesicles carrying signaling molecules; they’re cell-free and avoid some risks of live cell therapy.
Q: Will MFAT regrow cartilage?
MFAT can improve symptoms and provide regenerative signals; evidence for actual, durable cartilage regrowth in humans is limited but promising in certain series.
Q: Which therapy has the strongest RCT evidence?
PRP and some BMAC protocols have multiple RCTs; many newer options (exosomes, gene therapy, advanced scaffolds) are in early-phase human trials or strong preclinical work.
Final Thoughts
If your goal is to alter disease progression in early OA rather than only relieve pain, the most promising path likely involves multimodal regenerative strategies: a biologic payload optimized for anti-inflammatory and chondroinductive activity (exosome or cell product) plus a delivery system that enhances retention (scaffold, hydrogel), and — where available and appropriate — targeted gene delivery for sustained therapeutic expression. For routine practice today, MFAT and standardized BMAC offer pragmatic options with growing clinical evidence; exosomes, peptide-scaffolds and gene therapy are high-potential next steps that will enter broader clinical use as trials mature.